|
10/5/2023 0 Comments Entropy equation
When a hot object is placed in the room, it quickly spreads heat energy in all directions. When water in a dish is set on a counter, it eventually evaporates, the individual molecules spreading out in the surrounding air. For instance, if a car tire is punctured, air disperses in all directions. However, we see examples of entropy in our everyday lives. Therefore, any change that results in a higher temperature, more molecules, or a larger volume yields an increase in entropy.The meaning of entropy is difficult to grasp, as it may seem like an abstract concept. As well, increasing the volume of a substance increases the number of positions where each molecule could be, which increases the number of microstates. Increasing the number of molecules in a system also increases the number of microstates, as now there are more possible arrangements of the molecules. This increases the number of microstates possible for the system. We can estimate changes in entropy qualitatively for some simple processes using the definition of entropy discussed earlier and incorporating Boltzmann’s concept of microstates.Īs a substance is heated, it gains kinetic energy, resulting in increased molecular motion and a broader distribution of molecular speeds. A process that gives an increase in the number of microstates therefore increases the entropy. Microstates is a term used to describe the number of different possible arrangements of molecular position and kinetic energy at a particular thermodynamic state. Where k is the Boltzmann constant (1.38 × 10 −23 J/K), and W is the number of microstates. He developed an equation, known as the Boltzmann equation, which relates entropy to the number of microstates ( W): Ludwig Boltzmann (1844–1906) pioneered the concept that entropy could be calculated by examining the positions and energies of molecules. The Boltzmann Equation Figure 18.2 “Ludwig Boltzmann” Figure 18.1 “Two-Atom, Double-Flask Diagram.” When the stopcock is opened between the flasks, the two atoms can distribute in four possible ways. Thus we can say that it is entropically favoured for the gas to spontaneously expand and distribute between the two flasks, because the resulting increase in the number of possible arrangements is an increase in the randomness/disorder of the system. If we increased the number of atoms, we would see that the probability of finding all of the atoms in the original flask would decrease dramatically following (½) n, where n is the number of atoms. The likelihood of all atoms being found in their original flask, in this case, is only 1 in 4. If we were to take snapshots over time, we would see that these atoms can have four possible arrangements. When the stopcock is opened, both atoms are free to move around randomly in both flasks. At first, both atoms are contained in only the left flask. In this system, we have placed two atoms of gas, one green and one blue. The Molecular Interpretation of EntropyĬonsider the following system, where two flasks are sealed together and connected by a stopcock (see Figure 18.1 “Two-Atom, Double-Flask Diagram”). These definitions can seem a bit vague or unclear when you are first learning thermodynamics, but we will try to clear this up in the following subsections. 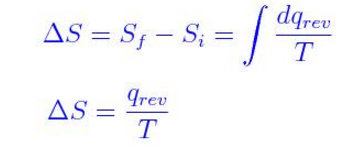
But what exactly is entropy? Entropy is typically defined as either the level of randomness (or disorder) of a system or a measure of the energy dispersal of the molecules in the system. 
The second law of thermodynamics states that a spontaneous process will increase the entropy of the universe. To assess the spontaneity of a process we must use a thermodynamic quantity known as entropy ( S). To be able to estimate change in entropy qualitatively.To gain an understanding of the Boltzmann equation and the term microstates.To gain an understanding of the term entropy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
